The main classes of inorganic compounds
Introduction. All known substances are divided into simple and complex. Simple
substances are divided into metals and non-metals. Today, the known 105 chemical
elements make up more than 30,000,000 compounds, of which about 300,000 form
inorganic compounds. Although each compound has its own unique properties, many
chemical compounds share similar properties with each other. Based on these
general properties, they divide inorganic compounds into oxides, acids, bases and salts [4, p. 98].
Oxides. Compounds of oxygen with other elements are called oxides. There
are only two kinds of atoms in oxides.
1.
NO2, SO2, H2O, CO2,
N2O5, NO, N2O are common non metal oxides, they
have covalent bond structure.
2.
Na2O, FeO, Al2O3, CaO,
SiO2, MgO, CuO, PbO are some common metal oxides they have ionic
structure.
Some oxides can react directly with water to form an acidic, basic,
or amphoteric solution. An amphoteric solution is a substance that
can chemically react as either acid or base. However, it is also possible for an
oxide to be neither acidic nor basic. There are different properties which help
distinguish between the three types of oxides. The term anhydride ("without water") refers to
compounds that assimilate H2O to form either an acid or a base upon
the addition of water.
Oxides are binary compounds of oxygen with another element, e.g.,
CO2, SO2, CaO, CO, ZnO, BaO, H2O, etc. These
are termed as oxides because here, oxygen is in combination with only one
element. Based on their acid-base characteristics oxides are classified as
acidic or basic. An oxide that combines with water to give an acid is termed as
an acidic oxide. The oxide that gives a base in water is known as a basic oxide
[5, p. 1].
Naming of Oxides. They are named like binary compounds.
MgO : Magnesium oxide
FeO : Iron (II) oxide
Na2O : Sodium oxide
SO2 : Sulfur dioxide
CO2 : Carbon dioxide
P2O5 : Diphosphorus pentoxide
SnO2 : Tin(IV) oxide
NO : Nitrogen monoxide
Classification of Oxides
A.
Neutral Oxides.
·
They are oxygen poor compounds of non metals.
·
They have neither acidic nor basic
properties.
·
They do not react with acids, bases and water. They are slightly
soluble in water.
·
CO, NO and N2O are neutral oxides
B.
Acidic Oxides. Oxygen rich compounds of non metals are called acidic oxides.
SO2, NO2, P2O5,
Cl2O7, CO2 are examples. Their solutions are
acidic.
They are known as acidic anhydrides. Acidic oxide + water → Acid
CO2 + H2O →
H2CO3
P2O5 + 3H2O →
2H3PO4
N2O5 + H2O →
2HNO3
Acidic oxides are, therefore, known as acid anhydrides, e.g., sulfur
dioxide is sulfurous anhydride; sulfur trioxide is sulfuric anhydride. When
these oxides combine with bases, they produce salts, e.g.
SO2+
2NaOH
→
H2SO3
+
H2O
C.
Basic Oxides. Generally Group 1 and Group 2 metal oxides are called basic oxides.
Na2O, CaO, Li2O, MgO, K2O are examples. Their
solutions are basic. They are known as basic anhydrides. Basic oxide + water →
Base
Na2O + H2O → 2NaOH
MgO + H2O → Mg(OH)2
K2O + H2O → 2KOH
These metallic oxides are therefore, known as basic anhydrides. They
react with acids to produce salts, e.g.
MgO + 2HCl→MgCl2 + H2O
Na2O + H2SO4 →
Na2SO4 + H2O
D.
Mixed Oxides. Compounds that contain two oxides of the same metal are called mixed
oxides. Fe3O4, Mn3O4,
Pb3O4 are examples.
They behave as if they are two separate oxides in chemical
reactions.
Fe3O4 :
FeO.Fe2O3 : Iron (II, III) oxide
Mn3O4 : MnO.Mn2O3 :
Manganese(II, III) oxide
Pb3O4 : 2PbO.PbO2 : Lead (II, IV)
oxide
E.
Amphoteric Oxides. Amphoteric oxides are metallic oxides, which show both basic as well
as acidic properties. When they react with an acid, they produce salt and water,
showing basic properties. While reacting with alkalies they form salt and water
showing acidic properties, e.g.
ZnO + 2HCl → ZnCl2 + H2O (basic
nature)
ZnO + 2NaOH → Na2ZnO2 + H2O (acidic
nature)
Al2O3 + 3H2SO4 →
Al2(SO4)3 + 3H2O (basic
nature)
Al2O3 + 2NaOH → 2NaAlO2 +
H2O (acidic nature)
Amphoteric oxides have both acidic and basic properties. A common
example of an amphoteric oxide is aluminum oxide. In general, amphoteric oxides
form with metalloids. (see chart below for more detail). Example with acidic
properties:
Al2O3 + H2O → 2Al(OH)3 +
2H+
Example with basic properties:
Al2O3 + H2O → 2Al3+ +
3OH−
F.
Peroxides and dioxides. Oxides: Group 1 metals react rapidly with oxygen to produce several
different ionic oxides, usually in the form of M2O. With the oyxgen
exhibiting an oxidation number of -2.
4Li + O2 → 2Li2O
Peroxides: Often Lithium and Sodium reacts with excess oxygen to
produce the peroxide, M2O2. with the oxidation number of
the oxygen equal to -1.
Compounds that contain O2-2 ion are called
peroxides: H2O2, K2O2,
Li2O2, CaO2, MgO2 are
examples.
H2 + O2 →
H2O2
Superoxides: Often Potassium, Rubidium, and Cesium react with excess
oxygen to produce the superoxide, MO2 with the oxidation number of
the oxygen equal to -1/2.
Cs + O2 → CsO2
A peroxide is a metallic oxide which gives hydrogen peroxide by the
action of dilute acids. They contain more oxygen than the corresponding basic
oxide, e.g., sodium, calcium and barium peroxides.
BaO2 + H2SO4 → BaSO4 +
H2O2
Na2O2 + H2SO4 →
Na2SO4 + H2O2
Dioxides like PbO2 and MnO2 also contain higher
percentage of oxygen like peroxides and have similar molecular formulae. These
oxides, however, do not give hydrogen peroxide by action with dilute acids.
Dioxides on reaction with concentrated HCl yield Cl2 and on reacting
with concentrated H2SO4 yield
O2.
PbO2 + 4HCl → PbCl2 + Cl2 +
2H2O
2PbO2 + 2H2SO4 → 2PbSO4 +
2H2O + O2
G.
Compound oxides. Compound oxides are metallic oxides that behave as if they are made
up of two oxides, one that has a lower oxidation and one with a higher oxidation
of the same metal, e.g.,
Red lead: Pb3O4 = PbO2 +
2PbO
Ferro-ferric oxide: Fe3O4=
Fe2O3 + FeO
On treatment with an acid, compound oxides give a mixture of
salts.
Fe3O4 + 8HCl → 2FeCl3 +
FeCl2 + 4H2O [5, p. 1]
Preparation of Oxides. Oxides can be generated via multiple reactions. Below are a
few.
1.
By direct heating of an element with oxygen Many metals and
non-metals burn rapidly when heated in oxygen or air, producing their oxides,
e.g.,
2Ca + O2 → 2CaO
P4 + 5O2 →
P2O5
2.
By reaction of oxygen with compounds at higher temperatures. At
higher temperatures, oxygen also reacts with many compounds forming oxides, e.g.
Sulphides are usually oxidized when heated with oxygen.
2PbS + 3O2 → 2PbO + 2SO2
2ZnS + 3O2 → 2ZnO + 2SO2
When heated with oxygen, compounds containing carbon and hydrogen are
oxidized.
C2H5OH + 3O2 → 2CO2 +
3H2O
By thermal decomposition of certain compounds like hydroxides,
carbonates, and nitrates
CaCO3 → CaO + CO2
Cu(OH)2 → CuO + H2O
3.
By oxidation of some metals with nitric acid
2Cu + 8HNO3 → 2CuO + 8NO2 + 4H2O +
O2
Sn + 4HNO3 →
SnO2 + 4NO2 + 2H2O
4.
By oxidation of some non-metals with nitric
acid
C + 4HNO3 → CO2 + 4NO2 +
2H2O [5, p. 4]
Chemical properties
1.
Interaction with water
Na2O + H2O → 2NaOH
SO3 + H2O →
2H2SO4
2.
Interaction with acid or base
MgO + H2SO4 → MgSO4 +
H2O
CuO + 2HCl → CuCl2 + H2O
CO2 + Ba(OH)2 → BaCO3 +
H2O
SO2 + 2NaOH → Na2SO3 +
H2O
ZnO + 2NaOH → Na2ZnO2 +
H2O
ZnO + H2SO4 → ZnSO4 +
H2O
ZnO + 2NaOH + H2O →
Na2[Zn(OH)4]
3.
Interaction of basic and acid oxide with each other leads to salt
formation
Na2O + CO2 →
Na2CO3
4.
Reduction up to simple substances:
3CuO + 2NH3 → 3Cu + N2 +
3H2O
P2O5 + 5C → 2P + 5CO
Salts are ionic compounds of anions and cations (figure2.1). NaCl,
CaCO3, ZnBr2, FeSO4, NH4Cl,
Mg(NO3)2, LiF, AlPO4 …etc are examples.
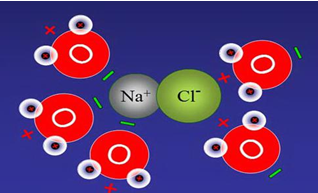
Figure 2.1 Structure of salt
Melting and boiling points: Salts are mostly solids which melt as well as boil at high
temperatures.
Solubility in water: Salts are generally soluble in water. For example, sodium chloride,
potassium sulphate, aluminium nitrate, ammonium carbonate, etc., are soluble
salts while silver chloride, lead chloride, copper carbonate, etc., are
insoluble in water.
Water of crystallization: Generally, salts are found as crystals with water molecules present
in them. This water is called water of crystallization and such salts are called
hydrated salts.
For example, copper sulphate crystal has five molecules of water for
each copper sulphate molecule. This is written as
CuSO4∙5H2O. This water of crystallization gives the
crystal its shape. It also gives colour to some crystals. On heating, hydrate
salts lose their water of crystallization and, as a result, the crystals lose
their shape and colour and change to a powdery substance.
The hydrated salts that have lost their water of crystallization are
called anhydrous
salts.
When hydrated copper sulphate is heated, it gives out water molecules
to form white powdery anhydrous copper sulphate. On addition of water, this
substance can convert back to a hydrated copper sulphate solution again [6, p,
1].
Naming of Salts
• In the naming of salts first metal ion (positive ion) then name of
negative ion is read.
NaCl: Sodium chloride
BaC2O4: Barium oxalate
FeCl2: Iron (II) chloride
KMnO4: Potassium permanganate
NH4Br: Ammonium bromide
PbI2: Lead (II) iodide [6, p. 1]
Classification of Salts
A.
Neutral Salts
• They are formed from the reactions of strong acids with strong
bases.
• They have neither acidic nor basic character.
• Their solutions are neutral.
• NaCl, LiNO3, KNO3, NaBr,
Li2SO4 …etc are examples.
B.
Acidic Salts
• They are formed from the reactions of strong acids with weak bases.
• They have acidic character.
• The salt still has hydrogen atom(s)
from an acid which can further be replaced by metallic ions. Examples include:
NaHSO4, NaHCO3 and NaHS
C.
Basic Salt
• They are formed from the reactions of weak acids with strong bases.
• They have basic character.
• The salt contains hydroxides
together with metallic ions and negative ions from an acid. Examples are basic
zinc chloride, ZnOHCl, basic magnesium chloride.
D.
Double
salt
Salt that ionizes to produce three different types of ions in
solution, two of these are usually positively charged and the other negatively
charged. Examples are ammonium iron(II) tetraoxosulphate(VI) hexahydrate,
(NH4)2 Fe(SO4)2 ×
6H2O; potash alum or aluminium potassium tetraoxosulphate(VI)
dodecahydrate, KAl(SO4)2 × 12H2O; and chrome
alum or chromium(III) potassium tetraoxosulphate(VI) dodecahydrate,
KCr(SO4)2 × 12H2O.
E.
Complex
salt
The salt contains complex ions, i.e. ions consisting of a charged
group of atoms. Examples are sodium
tetrahydroxozincate(II)
Na2Zn(OH)4 (aq) ↔ 2Na+ (aq) +
Zn(OH)42- (aq) [7, p. 1]
Preparation of Salts. Salts are formed by a chemical reaction between:
·
By the reaction between a base and an acid:
Cu(OH)2 + 2HCl → CuCl2 +
2H2O
·
By the reaction between metal and an acid:
Fe + 2HCl → FeCl2 + H2
·
By direct union of a metal and a
nonmetal:
Fe + S → FeS
·
A base and an acid anhydride, e.g.,
2NaOH + CrO3 → Na2CrO4 +
H2O
·
By the union an acid(or an acidic oxide) and a basic anhydride:
3CaO + P2O5 →
Ca3(PO4)2
·
By the reaction between a metal and a
base:
Zn + 2NaOH → Na2ZnO2 +
H2
3S + 6NaOH → 2Na2S + Na2SO3 +
3H2O
·
Salts can also form if solutions of different salts are mixed, their
ions recombine, and the new salt is insoluble and precipitates
(see: solubility equilibrium), for example:
Pb(NO3)2 + Na2SO4 →
PbSO4↓ +2NaNO3 [8, p. 1]
Chemical Properties of Salts
1.
Reaction with a metal: Salts can react with metals according to
activity strength.
Zn(s) + 2AgNO3 (aq) → 2Ag(s) + Zn(NO3)2
(aq)
2.
Reaction with a base: A salt reacts with a base to produce another
salt and base.
(NH4)2SO4 + 2NaOH →
Na2SO4 + 2NH4OH
3.
Reaction with an acid: When a salt reacts with an acid, another salt
and are formed.
NaHCO3 + H2SO4 → NaHSO4 +
H2CO3
4.
Behaviour of salts towards water: When a salt is dissolved in water,
the solution may be neutral, acidic or alkaline. This depends upon the nature of
the salt used.
a)
A normal salt derived from a strong base gives an alkaline
solution.
NaCl + 2H2O → NaHSO4 + HCl (at law
temperature)
2NaCl + H2O → Na2SO4 + 2HCl (at high
temperature)
b)
A normal salt derived from a weak acid and strong base gives an
alkaline solution.
Na2CO3 + 2H2O → 2NaOH +
CO2 + H2O
CH3COONa + H2O → CH3COOH +
NaOH
c)
A salt derived from a strong acid and weak base gives an acidic
solution.
AlCl3 + 3H2O → Al(OH)3 +
3HCl
NH4Cl + H2O → NH4OH +
HCl
d)
Solution of acidic salts are acidic to litmus, i.e., these solutions
turn blue litmus paper red [6, p. 1].
Bases. In chemistry, bases are substances that, in aqueous solution, are slippery to the touch, taste astringent, change the color of indicators (e.g., turn red litmus paper blue), react with acids to form salts, promote certain chemical reactions (base catalysis), accept protons from any proton donor, and/or contain completely or partially
displaceable OH− ions. Examples of bases are the hydroxides of the alkali metals and the alkaline earth metals (NaOH, Ca(OH)2, etc.).
Properties. Some general properties of bases include
- Slimy or soapy feel on fingers, due to saponification of the lipids in human skin.
- Concentrated or strong bases are caustic on organic matter and react violently with acidic substances.
- Aqueous solutions or molten bases dissociate in ions and conduct electricity.
- Reactions with indicators: bases turn red litmus paper blue, phenolphthalein pink, keep bromothymol blue in itsnatural colour of blue, and turns methyl orange yellow.
- The pH level of a basic solution is higher than 7.
- Bases are bitter in taste [9, p. 1].
Naming of Bases. The word “hydroxide” is added after the name of metal ion in the
naming of bases.
Mg(OH)2 :
Magnesium hydroxide
KOH : Potassium hydroxide
NaOH : Sodium hydroxide
Ba(OH)2 : Barium hydroxide
Cu(OH)2 : Copper (II) hydroxide
NH3 : Ammonia
Classification of Bases
Strong bases. A strong base is a basic chemical compound that is able to
deprotonate very weak acids in an acid-base reaction. Compounds with a pKa of
more than about 13 are called strong bases. Common examples of strong bases are
the hydroxides of alkali metals and alkaline earth metals like NaOH and
Ca(OH)2. Very strong bases are even able to deprotonate very weakly
acidic C-H groups in the absence of water. Hydroxide compounds in order of
strongest to weakest:
·
Potassium
hydroxide (KOH)
·
Barium
hydroxide (Ba(OH)2)
·
Cesium
hydroxide (CsOH)
·
Sodium
hydroxide (NaOH)
·
Strontium
hydroxide (Sr(OH)2)
·
Calcium
hydroxide (Ca(OH)2)
·
Lithium
hydroxide (LiOH)
·
Rubidium
hydroxide (RbOH)
The cations
of these strong bases appear in the 1st and 2nd groups of the periodic table
(alkali and earth alkali metals).
Superbases. Group 1 salts of carbanions, amides, and hydrides tend to be even
stronger bases due the conjugate acids, which are stable hydrocarbons, amines,
and water. Usually these bases are created by adding pure alkali metals such as
sodium into the conjugate acid. They are called superbases and it is not possible to keep them in water solution, due to
the fact they are stronger bases than the hydroxide ion and as such it will
deprotonate the conjugate acid water. For example the ethoxide ion (conjugate
base of ethanol) in the presence of water will undergo this reaction.
CH3CH2O- + H2O → CH3CH2OH + OH-
·
Butyl
lithium (n-BuLi)
·
Lithium
diisopropylamide
(C6H14LiN)
·
Sodium
amide (NaNH2)
·
Sodium
hydride (NaH)
Weak Base. A "Weak Base" is one that does not fully ionize in
solution. When a base ionizes, it takes up a hydrogen ion from the water around
it, leaving an OH- ion behind. Weak bases have a higher
H+ concentration than strong bases. Weak bases exist in chemical
equilibrium in the same way weak acids do. The Base Ionization Constant
Kb indicates the strength of the base. Large Kbs
belong to stronger bases. The pH of a base is greater than 7 (where 7 is the
neutral number; below 7 is an acid), normally up to 14. Common example of a weak
base is ammonia, which is used for cleaning.
Examples of Weak Bases:
·
Alanine (C3H5O2NH2)
·
Ammonia (water)
(NH3 (NH4OH))
·
Dimethylamine
((CH3)2NH)
·
Ethylamine
(C2H5NH2)
·
Glycine (C2H3O2NH2)
·
Hydrazine (N2H4)
·
Methylamine
(CH3NH2)
·
Trimethylamine ((CH3)3N) [9, p. 1]
Preparation of Bases
1.
Reactions
of active metals (alkaline and alkaline earth metals) with
water
2Na + 2H2O → 2NaOH +
H2
Ca + 2H2O →
Ca(OH)2 + H2
2.
Interaction oxides of active metals with water
BaO + H2O →
Ba(OH)2
3.
Electrolysis water solutions of salts
2NaCl + 2H2O → 2NaOH +
H2 + Cl2
Chemical Properties of Bases. Alkalis Insoluble bases
1.
Action to
indicators litmus – blue
methylorange – yellow phenolphthalein – crimson
2.
Interaction with acid oxides
2KOH + CO2 →
K2CO3 + H2O
KOH + CO2 →
KHCO3
3.
Interaction with acids (reaction of
neutralization)
NaOH + HNO3 →
NaNO3 + H2O
Cu(OH)2 + 2HCl →
CuCl2 + 2H2O
4.
Reaction
of exchange with salts
Ba(OH)2 +
K2SO4 → 2KOH + BaSO4↓
3KOH +
Fe(NO3)3 → Fe(OH)3↓ +
3KNO3
5.
Thermal
decomposition tоС
Cu(OH)2 → CuO +
H2O
Mg(OH)2 → MgO + H2O [10, p.
1]
Acids. The term acid was first used in the seventeenth century; it comes
from the Latin root ac-, meaning “sharp”, as in acetum, vinegar. Acids have long
been recognized as a distinctive class of compounds whose aqueous solutions
exhibit the following properties:
·
A characteristic sour taste;
·
ability to change the color of litmus1 from blue to red;
·
react with certain metals to produce gaseous H2;
·
react with bases to form a salt and water.
The first chemical definition of an acid turned out to be wrong: in
1787, Antoine Lavoisier, as part of his masterful classification of substances,
identified the known acids as a separate group of the “complex substances”
(compounds). Their special nature, he postulated, derived from the presence of
some common element that embodies the “acidity” principle, which he named
oxygen, derived from the Greek for “acid former”. Lavoisier had assigned this
name to the new gaseous element that Joseph Priestly had discovered a few years
earlier as the essential substance that supports combustion. Many combustion
products (oxides) do give acidic solutions, and oxygen is in fact present in
most acids, so Lavoisier’s mistake is understandable.
In 1811 Humphrey Davy showed that muriatic (hydrochloric) acid (which
Lavoisier had regarded as an element) does not contain oxygen, but this merely
convinced some that chlorine was not an element but an oxygen-containing
compound. Although a dozen oxygen-free acids had been discovered by 1830, it was
not until about 1840 that the hydrogen theory of acids became generally
accepted. By this time, the misnomer oxygen was too well established a name to
be changed.
Compounds dissolving in water by producing H+ ion are
called acids. Many of the fruits and vegetable contain acids; in lemon, apple,
tomatoes, orange, as well as in car batteries, and in cleaning
materials.
HCl(g) ® H+(aq) +
Cl-(aq)
H2SO4 ® 2H+(aq) +
SO4-2(aq)
HNO3 ® H+(aq) +
NO3-(aq)
CH3COOH ® H+(aq) +
CH3COO-(aq)
·
They have sour taste.
·
They change the color of litmus paper to
red.
·
Their aqueous solutions conduct
electricity.
·
They are corrosive substances.
·
Most of them are soluble in water.
Naming of
Acids
·
Acids containing two types of atoms are called binary acids.
·
Their names follow the form hydro + nonmetal name + acid.
HCl : Hydrochloric acid
HI : Hydroiodic acid
H2S : Hydrosulfuric acid
HF : Hydrofluoric acid
Acids containing oxygen atoms are called oxy acids. Their names
follow the form –ic + acid, or –ous + acid.
H3BO3 : Boric acid
H3PO4 : Phosphoric acid
H2SO4 : Sulfuric acid
H2SO3 : Sulfurous acid
HNO3 : Nitric acid
HNO2 : Nitrous acid
Classification of Acids
According to Strength. If an acid ionizes completely, it is an strong acid, and if it
ionizes partially it is a weak acid.
Strong acids: HCl, H2SO4, HNO3, HI,
HBr, HClO4
Weak acid: HF, H2SO3, HNO2,
H2S, H3PO4, CH3COOH, HCN,
H2CO3
According to Number of Hydrogen Atoms. According to number of H+ ion produced acids are classified as
monoprotic, diprotic or triprotic.
Monoprotic acids: HCl, HNO3, HI, HBr, HClO4
HCl(g) + H2O → H+(aq) +
Cl-(aq)
CH3COOH (l) + H2O → H+(aq) +
CH3COO-(aq)
Diprotic acids: H2SO3, H2S,
H2CO3, H2SO4
H2CO3(l) + H2O → 2H+(aq)
+ CO3-2(aq)
Triprotic acids: H3PO4, H3AsO4
H3AsO4(s) + H2O → 3H+
(aq) + AsO4-3(aq) [11, p. 1]
Preparation of Acids
1.
Some
acids can be prepared by the direct combination of a non-metal with hydrogen.
For e.g.,
H2(g) + Cl2(g) → 2HCl(g)
H2(g) + I2(s) → 2HI(g)
H2(g) + S(l) → 2H2S(g)
In the above reactions, the obtained gases like hydrogen chloride,
hydrogen iodide and hydrogen sulphide are also called the acid anhydrides. They
exhibit acidic properties, only when dissolved in water.Therefore an acid
anhydride is a substance that dissolves in water to form an acid. In other
words, it is the acid minus water (acid - water = acid anhydride).
Examples: CO2 gas is
the acid anhydride of carbonic acid, SO3 gas is the acid
anhydride of carbonic acid, HCl gas is the acid anhydride of hydrochloric
acid.
H2O(l) + CO2(g) →
H2CO3(aq)
H2O(l) + SO3(s) →
H2SO4(aq)
H2O(l) + HCl(g) →
H2CO3(aq)
2.
Preparation by dissolving acidic oxide in water. Oxides that can add on hydrogen ions to their
molecules are called acidic oxides. Thus when they dissolve in water they
associate with hydrogen ions to form acids.
6H2O(l) + 2P2O5(s) →
4H3PO4(aq)
3.
Preparation by the displacement of metal ions of salts of volatile
acids by less volatile acid. The hydrogen ions
of less volatile acids, like concentrated sulphuric acid, can help displace
metal ions of salts of volatile acids, to produce the
acid.
NaCl (s) + H2SO4 → NaHSO4 (aq) +
HCl(g)
KNO3(s) + H2SO4 → KHSO4
(aq) + HNO3 (aq)
4.
Preparation by the action of dilute acids on
salts
Na2CO3 (aq) + 2HCl (aq) → 2NaCl (aq) + CO2
(g) + H2O (l)
FeS(s) + H2SO4 → FeSO4 (aq) +
H2O (l)
Hydrogen sulphide is the acid anhydride.
5.
Preparation by the oxidation of non-metals by concentrated nitric
acid. Non metals like sulphur, phosphorous get
oxidized by concentrated nitric acid to form their respective
acids.
S(s) + 6HNO3 (aq) → H2SO4 (aq) +
6NO2 (g) + 2H2O (l)
P(s) + 5HNO3 (aq) → H3PO4 (aq) +
5NO2 (g) + H2O (l) [12, p. 1]
Chemical Properties of Acids
1.
Action to indicators
Litmus – red
Methylorange – pink
2.
Interaction with bases (reaction of
neutralization)
H2SO4 + 2KOH → K2SO4 +
2H2O
2HNO3 + Ca(OH)2 →
Ca(NO3)2 + 2H2O
3.
Interaction with basic oxides
CuO + 2HNO3 → K2SO4 +
2H2O
4.
Interaction with metals
Zn + 2HCl → ZnCl2 + H2
2Al + 6HCl → 2AlCl3 + 3H2
5.
Interaction with salts (reactions of exchange) at which stands out
gas or formed residual
H2SO4 + BaCl2 → BaSO4 +
2HCl
2HCl + K2CO3 → 2KCl + H2O + CO2
[10, p. 1]
Amphoteric Compounds
·
Most of the compounds of Zn, Al, Cr, Sn, Pb, and Be are amphoteric
compounds.
·
Oxides and hydroxides of these metals have both acidic and basic
characters.
·
They are in soluble in water and do not react with it.
·
ZnO, Al2O3, SnO, BeO,
Cr2O3, PbO are oxides, and Zn(OH)2,
Al(OH)3, Be(OH)2, Sn(OH)2, Pb(OH)2,
Cr(OH)3 are hydroxides.
ZnO + 2HCl → ZnCl2 + H2O
ZnO + 2NaOH → Na2ZnO2 +
2H2O